
The name under this image reads “Simple cubic. The first three images are in a row and are labeled “Lattice point locations” while the second three images are in a row labeled “Cubic unit cells.” The first image in the top row shows a cube with black dots at each corner while the first image in the second row is composed of eight spheres that are stacked together to form a cube and dots at the center of each sphere are connected to form a cube shape. Examples of crystal lattices include sodium chloride, diamond, quartz, graphite, zinc sulfide, ice, copper, silicon, potassium chloride, and aluminum.\): Cubic unit cells of metals show (in the upper figures) the locations of lattice points and (in the lower figures) metal atoms located in the unit cell. Lattice points are the points in a crystal lattice where the particles are located, and the position of these points determines the crystal structure. A set of visualizations developed for the purpose of explaining solid state lattice structures is presented in this paper. There are 14 types of crystal lattices, classified into seven different systems based on their symmetry. In conclusion, a crystal lattice is a fundamental concept in chemistry that describes the repeating pattern of particles in a crystalline solid. Aluminum: Aluminum has a face-centered cubic lattice structure. 
Potassium chloride: Potassium chloride has a face-centered cubic lattice structure.Silicon: Silicon has a diamond lattice structure. 
This particular particle may be an atom, a molecule or even ions These lattice points of a crystal are joined together by straight lines. Each point on the lattice represents one particle of the crystal, This is a lattice point. Copper: Copper has a face-centered cubic lattice structure. The fourteen Bravais Lattices show some similar characteristics.Ice: Ice has a hexagonal lattice structure.Zinc sulfide: Zinc sulfide has a zincblende lattice structure.Graphite: Graphite has a hexagonal lattice structure.Quartz: Quartz has a hexagonal lattice structure.Diamond: Diamond has a cubic lattice structure.Sodium chloride: Sodium chloride, also known as table salt, has a face-centered cubic lattice structure.There are different types of crystal lattice, which are given below: The position of the lattice points determines the crystal structure. The visualizations provide students with the option to view stacking patterns, extended structures, and individual unit cells for simple, body-centered and face-centered cubic, and hexagonal close-packed structures. These points are the intersections of the lattice lines, which represent the paths between the particles. A set of visualizations developed for the purpose of explaining solid state lattice structures is presented in this paper. Hexagonal system: In this system, the lattice points form a hexagonal prism.Ī lattice point is a point in a crystal lattice where the particles are located.Rhombohedral system: In this system, the lattice points form a parallelepiped, but the angles between the sides are all equal, and the sides are not perpendicular to each other.In ionic crystals, this defect forms when oppositely charged ions leave their lattice sites and become incorporated for instance at the surface, creating oppositely charged vacancies. Triclinic system: In this system, the lattice points form a parallelepiped, but the angles between the sides are all different. A Schottky defect is an excitation of the site occupations in a crystal lattice leading to point defects named after Walter H.Monoclinic system: In this system, the lattice points form a parallelepiped, but the angles between the sides are not equal. 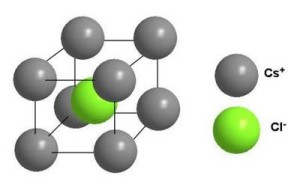
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
